制药过程的每一步都必须记录在案,这已不是什么秘密。然而,对于制药商来说,管理纸质文档可能非常耗时,而且人为错误也比较常见。
因此,制药公司决定实施所谓的电子批次记录 (EBR),以简化工作流程,最大限度地减少错误发生的可能性。本文将介绍 ANT 系统如何在制药行业实现从批量到包装流程的文档创建。
EBR 是什么?
制药行业的公司决定在生产中采用新的解决方案,他们关心的不仅仅是表格的数字化。EBR 解决方案还旨在最大限度地缩短填写表格字段所需的时间,并通过直接从机器上收集数据实现自动填表。表格上的数据可能包括:产品称重、混合、造粒、制表、涂层、包装和质量控制。直接从衡器、pH 值和电导率仪等设备收集数据通常也是一项要求。此外,公司还希望避免错误并跟踪生产过程中出现的偏差。
ANT 系统的实施不仅带来了高效的文档数字化。操作员在整个生产流程中都能得到指导,包括转换、质量、物流,当然还有维护流程。该系统符合 GMP 标准。
这使得准备或修改表格变得更加容易。不过,这仍然需要授权人员--只有拥有正确访问权限的人员才能对表格进行修改。所有这些都是为了防止错误或不当处理。
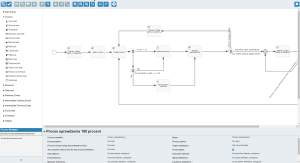
ANT 解决方案能为制药业带来什么?
ANT 提供完整的系统,包括机器数据采集、硬件、ERP 集成和基础设施。该系统还包括 MBR 模板创建。有了主批次记录(MBR),客户就可以按照正确的顺序设置系统中的所有活动,并为每个角色设定责任,从而创建 MBR 模板。每项活动都可以配置报告数据类型,如数字、文本、从自动化读取的数据等。然后可以根据系统中设置的质量限制对这些值进行验证。
已实施的表单字段的几个例子包括是否已执行操作。此外,还有一个产值字段和关于何时开始或停止某项具体任务的信息,以及对该任务所花费时间的计算。此外,还在表格上添加了电子签名和负责人的注释或备注。
ANT 系统还能自动收集生产阶段的工艺参数数据。其中包括
称重 - 梅特勒-托利多 - 称重数据和 Gareco 本地协议
混合 - 西门子 TCP
制粒机 - Siemens TCP
工作台 - IMA Kilian - B&R OPC UA
涂层 - 格拉特 - 西门子 TCP
吸塑包装生产线 - IMA,包括吸塑机 - OPC、串联机 - 数字 I/O、装箱机 - 以太网 TCP。
通过实施该解决方案,每个工作中心都配备了操作员面板,白色区域有不同的型号,灰色区域则根据需要有所不同。操作员面板上执行的一些操作包括选择生产订单、确认已完成/未完成操作、电子签名、逐步指导、显示文档、生产进度监控、可追溯性和 OEE 分析。
可通过 WEB 服务与 SAP 集成。ANT 主数据模块可对生产流程(转换矩阵、质量控制限制等)进行补充说明。
制药行业的无纸化流程是怎样的?
无纸化流程考虑到以数字方式指导操作员完成生产的特定部分,首先是活动显示--生产的待办事项列表。它还包括为每个生产步骤提供专用文档(图像、文档、视频)的转换信息。生产过程中的所有纸质文件都通过 ANT Factory Forms 实现了数字化。
借助 ANT Factory Forms,所有表单、表格和报告都能根据当前班次收集到的数据自动生成。当任务必须完成时,系统会及时监控并通知操作员。任何更改和偏差都会受到审计跟踪监控。
移动条形码扫描仪可对材料进行验证和跟踪。称重和包装过程需要扫描并确认物料是否可用于当前的生产订单,从而在 WIP 上建立可追溯性和当前物料库存。系统支持包装箱或容器标签打印和跟踪。
测试样品时,系统本身会在需要创建新样品时自动通知操作员。它还会通知操作员准备打印 ANT 制作的新标签。随后,实验室将对该标签进行扫描,并在工厂内执行操作。ANT 系统读取检测设备的数值,并自动将结果与设定的质量公差进行比较。系统会判断样品正确与否,并将信息反馈到操作面板。
每个批次都必须通过 ANT 系统的审批路径才能放行。这与工作流程因素有关。每个步骤都必须由通知公司特定角色的人员签署电子签名。如果工作流程中的当前步骤分配给了特定角色,就会发送通知。拥有适当权限的人员可以查看批量发布流程的停止位置,并监控完成每个步骤所需的 KPI。
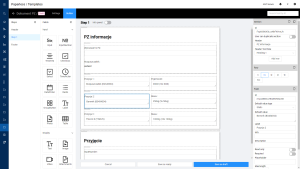
利用 EBR 进行数字记录管理
总之,上述所有因素使得 EBR 能够发挥作用。这使得整个生产流程的数字化文档管理成为可能。该系统可确保在各步骤中完整填写所有所需数据。EBR 创建的结果是一个包含完整报告和签名的 PDF 文件。
这对制药企业有什么好处?
首先,由于采用了工作流程和自动数据收集功能,该系统可缩短 35% 的批量放行时间。此外,由于采用了材料验证、定期质量检查和分步数字指导,还减少了 15% 的错误和浪费。此外,OTIF 也提高了 7%。
EBR 的实施还确保了公司各部门之间更快的沟通,减少了生产过程中的纸张消耗,并提供了对当前批次发布状态的全天候监控。
ANT 为您的制药企业提供所需的解决方案,使工作流程更高效、更环保。如需更多个性化建议,请随时联系我们的团队成员。




